Cancer therapy is in a constant state of development and new cancer drugs and surgical techniques are approved in a daily basis. The greatest misconception regarding cancer is that it is curable by a single medication, while the reality is a bit more realistic -and disappointing-. Cancers are a heterogenous group of diseases that only share the general mechanism of their occurrence, but differ in the way they present, the damage they produce, and the risk factors that produce them. Generally speaking, cancers are treated by surgery, chemotherapy, radiotherapy, and immunotherapy. Solid tumors are usually resected by surgery and then the remains are destroyed by chemotherapy or radiotherapy. Other cancers like blood cancers can’t benefit from surgery except in a minority of cases and is, therefore, mainly treated by chemotherapy and radiotherapy.
The main disadvantage of most conventional therapies is that they are hardly selective and thus, normal cells are damaged along with cancer cells leading to most side effects seen in chemotherapy and radiotherapy. This can be largely avoided by using a novel method called targeted therapy. Targeted therapies aim to exclusively target cancer cells as much as possible, minimizing the damage to normal cells and ensuring a better outcome in cancer patients. Among those targeted therapies is pembrolizumab.
What is pembrolizumab?

Pembrolizumab is a synthesized antibody. Antibodies are proteins produced in our bodies that attack a certain protein structure whether it belongs to a bacteria, a virus, or a cancer cell. They bind to such proteins, render the intruder immobile and activate immunity which destroys it. Pembrolizumab is therefore a protein which is artificially produced in a way to avoid it being allergic to humans, a procedure called humanization. It is a monoclonal antibody which means that it is targeted towards a certain protein structure called an antigen.
Pembrolizumab was created by Gregory Carven, Hans van Eenennaam, and John Dulos at Organon in 2006, and was approved by the Food and Drug Administration in 2014 for the treatment of advanced cases of Melanoma.
How does it work?
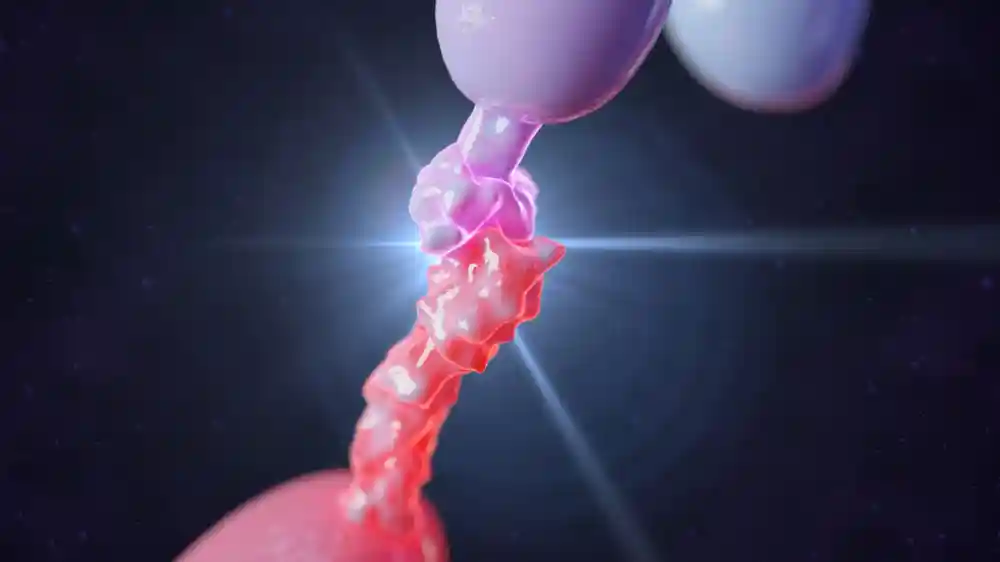
Our immunity is complex, and our white cells are the main soldiers when fighting infection and cancer. T-cells are a class of white blood cells which fight viruses, cancers, and mediate autoimmunity. They are very powerful and are, therefore, strictly regulated by the body. One of the mechanisms by which they are regulated in the presence of a protein called PD-1 which is short for programmed cell death receptor-1. This receptor is, under normal conditions, inhibited by another protein called PD-L1. Such interaction ensures that T-cells don’t attack body cells under normal conditions or during inflammation. This interaction is inhibited when the body detects a cancer cell, leading to the activation of T-cells and the destruction of the cancerous cell.
Tumor cells can overcome this if they express the PD-L1 receptors on their surface, preventing T-cells from attacking them. The mechanism of pembrolizumab is binding to the PD1 receptor on T-cells, preventing the interaction with the PD-L1 of cancer cells and allowing T-cells to attack them.
Uses

Due to its general mechanism of action, it can theoretically be used in a wide variety of cancer. Its mechanism of action doesn’t depend on the extent of the tumor or even its tissue of origin, but rather on some genetic abnormalities that are present at the center of the cancer cell. Several uses have already been approved since its conception and more are expected to come as clinical trials are still underway. Approved uses of pembrolizumab include:
- Surgically unresectable melanomas: Its first use, and in such condition, it is used alone. It can also be used following surgery in resectable cases.
- Non-small cell lung cancer: It is the most common type of lung cancer. This type of lung cancer is mostly resistant to chemotherapy. Pembrolizumab, however, can be used in cases which have certain mutations -genetic abnormalities- like PD1 receptors. It is used in cases in whose surgical resection is deemed impossible either because of disease progression or the general condition of the patient. It can be used alone or in combination with other drugs like carboplatin and paclitaxel.
- Small cell lung cancer: Small cell lung cancer forms about 15% of lung cancers and is considered more sensitive to chemotherapy than the non-small cell subtype; however, if chemotherapy proves ineffective, pembrolizumab is initiated as a monotherapy until a response occurs or toxicity by the drug ensues.
- Hodgkin’s lymphoma: Pembrolizumab is not considered a classical drug for Hodgkin’s lymphoma, but it is used in cases which relapsed after several lines of therapy.
- Squamous cell carcinoma: Squamous cell carcinoma is the second most common skin cancer and unlike basal cell carcinoma -the first-, it tends to spread to local lymph nodes and, in late stages- everywhere in the body. Pembrolizumab can be used alone in some cases of advanced and metastasized squamous cell carcinoma especially those of the head and neck or in a combination with chemotherapy like fluorouracil.
- Recurrent cancers or those that metastasized: Pembrolizumab is advocated for many cancers that are resistant to their usual lines of therapy like surgery or chemotherapy or those which, at the time of their discovery, had progressed beyond surgery and conventional therapy. They include cervical cancer, stomach cancer, liver cancer, esophageal cancer, and endometrial cancer. Here, we can see the main advantage of pembrolizumab in that its effect isn’t dependent on the type of tissue. In most of those cases, it is used alone.
Method of administration
Pembrolizumab is given intravenously either by slow injection or infusion.
Side effects of pembrolizumab

All drugs have side effects even the most widely taken and prescribed paracetamol is not completely innocent. There is a common misconception regarding the novel targeted cancer therapy that it is completely harmless which is never the case. It is true that this therapy is less destructive than the traditional chemotherapy, but it still acts through a blind activation of immunity in the case of pembrolizumab causing side effects from organ damage by the body’s immunity similar to those we see in autoimmune diseases as well as other side effects from the route of administration. Common side effects of pembrolizumab include:
- General side effects: They include nausea, vomiting, diarrhea and a general sense of weakness and easy fatiguability.
- Infusion reactions: Many intravenous medications have some form of an infusion reaction. The severity of the reaction depends on the medication’s chemistry and other individual factors in cases of allergy. Pembrolizumab commonly causes local inflammation at the site of injection which manifests as redness, edema, itchiness and joint pain. Some allergic reactions can, however, be severe in some cases that they become life-threatening. They are, however, expected and doctors are ready to deal with them when they appear.
- Inflammation at the site of action: According to the site of cancer, inflammation can be severe even in organs not affected by the cancer in some cases. Examples include:
- Inflammation of the thyroid gland: Such inflammation can cause a rise or fall of thyroid hormones (hyperthyroidism or hypothyroidism). This is most pronounced when pembrolizumab is used in head and neck skin cancer. The rise of thyroid hormone is due to the destruction of thyroid gland acini and the release of the stored hormones. This is temporary and a sure drop of function follows if destruction is extensive.
- Inflammation of the pancreas (pancreatitis): Which causes an increased blood glucose level due to a fall in insulin levels and if it is severe enough, permanent diabetes mellitus can occur.
- Inflammation of the colon and stomach: Which are usually not severe and resolve without problems.
- Inflammation of the liver (hepatitis): This presents either without symptoms with only a spike of liver enzymes or can become full-blown with jaundice and increased bleeding tendency due to decreased coagulation factors.
- Inflammation of the lungs: It occurs when pembrolizumab is used for the management of lung cancer and can cause life-threatening pneumonia especially if the patient is immunocompromised,
- Anemia: The autoimmune-like effect that ensues in some cases can, unfortunately, affect red cells. Anemia occurs especially when the drug is used for extensive periods of time like in the case of Hodgkin’s lymphoma where it can be used for up to 2 years. Anemia is unlikely to be severe enough to need a blood transfusion, but its management is challenging.
- Other non-specific symptoms include:
- Cough
- Fever
- Decreased weight
- Loss of appetite
- Muscle pain
- Breathlessness
- Constipation
- Diarrhea
- Swallowing difficulties
- High blood pressure
- Headache
Is pembrolizumab safe for pregnant women?

Drugs are assigned letters depending on their expected harm to a human fetus. This has to be proven by clinical trials. The assigned letters are A, B, C, D, and X. Category X shows that a drug has proven fetal damage in humans or animals and is, therefore, completely contraindicated in pregnancy.
Pembrolizumab has not been tested extensively, but its mechanism of action shows that it has potential harm to fetuses ranging from mild anomalies to fetal loss due to its chemical structure since it can cross the placenta and act on the fetus. Women are advised to use contraception for the period of therapy and at least 4 months after its termination. It may also be secreted in breast milk, so mothers are advised to discontinue breastfeeding during the duration of treatment and 4 months afterward.
